The recent tragic deaths linked to for-profit plasma donation clinics in Canada have ignited a critical global conversation about donor safety, particularly concerning the frequency with which individuals can safely donate this vital blood component. While plasma donation is a cornerstone of modern medicine, providing essential therapies for a myriad of life-threatening conditions, these incidents underscore the urgent need for rigorous oversight and harmonized international standards. The global demand for plasma-derived medicinal products (PDMPs) continues to rise, making the integrity and safety of the supply chain, from donor to patient, an issue of paramount importance for public health authorities, medical professionals, and humanitarian organizations worldwide. This renewed scrutiny compels a deeper examination of current practices, particularly in regions where financial incentives might influence donation frequency, potentially compromising donor well-being.
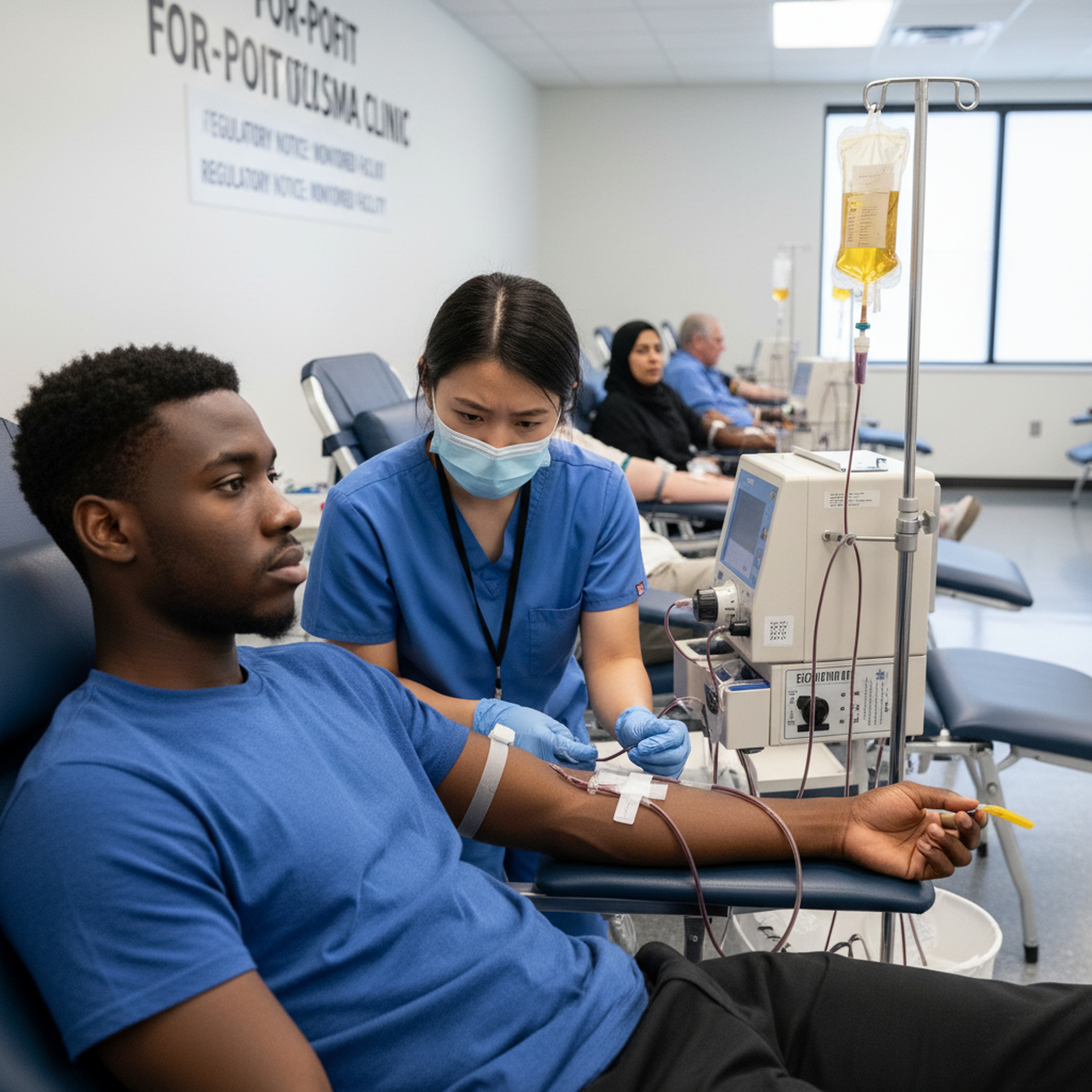
Plasma, the clear, yellowish liquid portion of blood, constitutes about 55% of total blood volume. It carries proteins, enzymes, antibodies, and other vital components throughout the body. Donated plasma is fractionated to produce a range of life-saving therapies, including immunoglobulins for immune deficiencies, albumin for burn victims and liver disease, clotting factors for hemophilia, and alpha-1 antitrypsin for genetic lung conditions. The process of donating plasma, known as plasmapheresis, involves drawing blood, separating the plasma, and returning the remaining blood components (red blood cells, white blood cells, platelets) to the donor. This procedure typically takes between 45 minutes to an hour and a half.
Under controlled conditions, plasma donation is generally considered safe. Donors undergo medical screening, including health questionnaires, blood tests, and vital sign checks, to ensure they meet eligibility criteria and are healthy enough to donate. Common side effects are usually mild and temporary, such as lightheadedness, fatigue, bruising at the needle site, or a tingling sensation. However, the human body requires time to replenish the proteins and fluids lost during donation. Frequent donations, especially those exceeding recommended guidelines, can lead to more serious health complications. These include significant protein depletion, electrolyte imbalances, dehydration, and a weakened immune response, potentially placing undue strain on the cardiovascular system and other vital organs.
The incidents in Canada, where individuals reportedly donated plasma as often as twice a week, highlight a critical vulnerability in systems that prioritize volume without adequate consideration for donor recovery. While some regulatory bodies permit donation twice weekly, this frequency is often contingent on stringent health monitoring and specific national guidelines designed to mitigate risk. The concern arises when for-profit models, driven by the need to meet high demand, might inadvertently create an environment where donors are incentivized to donate more frequently than is physiologically optimal, or where oversight might not be as robust as in voluntary, non-remunerated systems. These events serve as a stark reminder that even seemingly routine medical procedures carry inherent risks if not managed with the utmost care and adherence to best practices.
The regulatory landscape for plasma donation varies significantly across the globe. In many European countries and parts of Canada, plasma donation is largely based on a voluntary, non-remunerated model, often with stricter limits on donation frequency (e.g., once every two weeks or less). These systems typically prioritize donor health and safety above all else, viewing blood and plasma as a public good. In contrast, countries like the United States rely heavily on a remunerated model, where donors receive compensation for their plasma. This model, while crucial for meeting the substantial global demand for PDMPs, often permits more frequent donations, sometimes up to twice a week, under the premise of rigorous medical supervision. The recent Canadian deaths, occurring in clinics operating under a for-profit model similar to those prevalent in the US, have intensified calls for a re-evaluation of these differing regulatory approaches and their impact on donor welfare.
The ethical debate surrounding remunerated plasma donation is complex. Proponents argue that compensation incentivizes donation, ensuring a stable and sufficient supply of life-saving therapies. They also contend that with proper medical screening and monitoring, frequent donation can be safe. Critics, however, raise concerns about the potential exploitation of vulnerable populations who may feel compelled to donate more frequently than is advisable due for financial necessity, potentially overlooking or downplaying health risks. This creates a moral hazard where the pursuit of income might override personal health considerations, especially in socio-economically disadvantaged communities. Ensuring informed consent in such contexts becomes even more critical, requiring donors to fully understand the risks and benefits without undue financial pressure.
The global supply chain for plasma-derived medicinal products is highly interconnected. Plasma collected in one country is often processed and manufactured into therapies that are then distributed worldwide. The reliance of many nations on plasma imported from countries with remunerated donation systems means that donor safety practices in one region can have far-reaching implications for global health security. Any compromise in donor safety standards, or any incident that erodes public trust in the donation process, could destabilize this critical supply chain, potentially leading to shortages of essential medicines for patients globally. This interdependence necessitates a collective responsibility to uphold the highest standards of safety and ethics across all donation models.
In light of these recent events, there is a compelling argument for greater international collaboration to harmonize plasma donation safety standards. This includes developing globally recognized best practices for donor screening, monitoring, and frequency limits, irrespective of whether donation is voluntary or remunerated. Regulatory bodies worldwide should review their existing guidelines, particularly concerning the maximum permissible donation frequency and the robustness of health checks for frequent donors. Enhanced oversight of for-profit clinics is crucial, ensuring that profit motives do not overshadow the fundamental commitment to donor health. This could involve more frequent audits, stricter licensing requirements, and independent monitoring mechanisms to ensure compliance with safety protocols.
Public health organizations and advocacy groups play a pivotal role in this ongoing dialogue. They can educate potential donors about the risks and benefits of plasma donation, advocate for stronger regulatory frameworks, and support research into the long-term health effects of frequent donations. By fostering transparency and accountability within the plasma industry, these organizations contribute significantly to building and maintaining public trust, which is essential for the continued success of plasma collection programs. Nivaran Foundation, through its global news desk, aims to shed light on such critical health issues, advocating for policies that prioritize human well-being above all else.
The tragic incidents in Canada serve as a sobering reminder that while plasma donation is an invaluable act of altruism and a medical necessity, it must be conducted within a framework that unequivocally prioritizes donor safety. As the global demand for plasma therapies continues to grow, the imperative to ensure a safe, ethical, and sustainable supply becomes ever more pressing. This requires a concerted global effort to review and strengthen regulatory frameworks, enhance oversight, and foster a culture where the health and well-being of every donor are paramount. Only through such dedicated commitment can the world continue to harness the life-saving potential of plasma while safeguarding those who generously contribute to its supply.
Support Nivaran Foundation's mission to advocate for global health safety and ethical medical practices worldwide.
Support this work